 A new method that analyzes how individual immune cells react to the bacteria that cause the disease could pave the way for new vaccine strategies, and provide insights into fighting other infectious diseases around the world.
A new method that analyzes how individual immune cells react to the bacteria that cause the disease could pave the way for new vaccine strategies, and provide insights into fighting other infectious diseases around the world.
News
Koretzky and August elected to AAI leadership
 Two members of the Cornell Center for Immunology have been elected to leadership roles within the American Association of Immunologists.
Two members of the Cornell Center for Immunology have been elected to leadership roles within the American Association of Immunologists.
Dr. Gary Koretzky was named AAI President for 2021-2022. In his first Presidents Message, Dr. Koretzky outlines his vision to advance the association’s mission through advocacy and public engagement, member and trainee support and educational programs, and more. Dr. Koretzky serves as the Cornell Center for Immunology’s Director, and the Vice Provost for Academic Integration at Cornell University, and a Professor of Medicine at Weill Cornell Medicine.
Dr. Avery August was appointed an Councilor for 2021-2025. Dr. August serves on the Cornell Center for Immunology’s Executive Committee and is a Vice Provost for Academic Affairs, HHMI Professor and Professor of Immunology with in the department of Microbiology and Immunology at Cornell University’s College of Veterinary Medicine.
Drs. Koretzky and August will serve on the AAI Council which is composed of eight scientists elected by voting AAI members. Forward-looking in their determination to answer the significant questions facing scientists, Council-members are charged to speak on behalf of the AAI membership and act in the best interests of AAI. These leaders of AAI are recognized experts in their specific fields and experienced administrators. They are, above all things, passionate about science and the belief that good science is critical in overcoming so many of the challenges we face today.
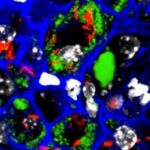
Geographic differences in gut microbiota boost immunity.
 Cornell researchers “humanized” mice with microbiota from three global populations and found that microbial differences alone can impact immune responses.
Cornell researchers “humanized” mice with microbiota from three global populations and found that microbial differences alone can impact immune responses.
Cancer vaccine improves outcomes in Lynch syndrome model
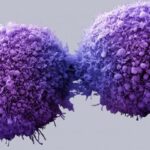 A new strategy for developing vaccines against cancer showed promise in a proof-of-concept study led by scientists at Weill Cornell Medicine, NewYork-Presbyterian and Heidelberg University Hospital.
A new strategy for developing vaccines against cancer showed promise in a proof-of-concept study led by scientists at Weill Cornell Medicine, NewYork-Presbyterian and Heidelberg University Hospital.
COVID-19 vaccines show safety, immunity in infant model
A group of scientists led by researchers at the University of North Carolina at Chapel Hill, Weill Cornell Medicine and NewYork-Presbyterian reported that the Moderna mRNA vaccine and a protein-based vaccine candidate elicited durable neutralizing antibody responses to SARS-CoV-2 in preclinical research. There were no adverse effects.
SUNY chancellor recognizes faculty and staff with excellence awards
 Two Center members have been selected for the 2020–21 State University of New York (SUNY) Chancellor’s Awards for Excellence. Brian Rudd, associate professor in the Department of Microbiology and Immunology, was a winner of the Chancellor’s Award for Excellence in Research and Scholarly Activity. John Parker, Ph.D. ’99, associate professor of virology at the Baker Institute for Animal Health, was a winner of the Chancellor’s Award for Excellence in Faculty Service.
Two Center members have been selected for the 2020–21 State University of New York (SUNY) Chancellor’s Awards for Excellence. Brian Rudd, associate professor in the Department of Microbiology and Immunology, was a winner of the Chancellor’s Award for Excellence in Research and Scholarly Activity. John Parker, Ph.D. ’99, associate professor of virology at the Baker Institute for Animal Health, was a winner of the Chancellor’s Award for Excellence in Faculty Service.
Better implant device may ease therapy for Type 1 diabetes
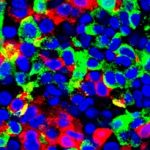 A Cornell-led research team’s improved cell therapy device effectively secreted insulin and controlled blood sugar in diabetic mice for up to six months – showing promise for the possibility of an effective, complication-free treatment for Type 1 diabetes, a chronic disease with no known cure.
A Cornell-led research team’s improved cell therapy device effectively secreted insulin and controlled blood sugar in diabetic mice for up to six months – showing promise for the possibility of an effective, complication-free treatment for Type 1 diabetes, a chronic disease with no known cure.
Award commends typhoid toxin and potential peptide therapy research
 Graduate students and postdoctoral researchers in the Department of Microbiology and Immunology faced stiff competition for this year’s Biocytogen Best Research Paper Award. Their work in infectious disease and host immune responses has been recognized in peer-reviewed journals, and faculty members faced the difficult task of selecting the winning entries. First- and second-place winners of this year’s award are Drs. Sohyoung Lee and Abrrey Monreal, respectively.
Graduate students and postdoctoral researchers in the Department of Microbiology and Immunology faced stiff competition for this year’s Biocytogen Best Research Paper Award. Their work in infectious disease and host immune responses has been recognized in peer-reviewed journals, and faculty members faced the difficult task of selecting the winning entries. First- and second-place winners of this year’s award are Drs. Sohyoung Lee and Abrrey Monreal, respectively.
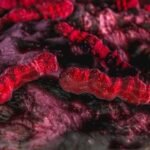
Study finds ‘Achilles heel’ of Crohn’s-linked bacteria
 The discovery of an “Achilles heel” in a type of gut bacteria that causes intestinal inflammation in patients with Crohn’s disease may lead to more targeted therapies for the difficult-to-treat disease, researchers have found.
The discovery of an “Achilles heel” in a type of gut bacteria that causes intestinal inflammation in patients with Crohn’s disease may lead to more targeted therapies for the difficult-to-treat disease, researchers have found.
Fungi in the gut prime immunity against infection
 Common fungi, often present in the gut, teach the immune system how to respond to their more dangerous relatives, according to new research from scientists at Weill Cornell Medicine. A study, published Feb. 5 in Cell, reveals a new twist in the complex relationship between humans and their associated microbes, and points the way toward novel therapies that could help combat a rising tide of drug-resistant pathogens.
Common fungi, often present in the gut, teach the immune system how to respond to their more dangerous relatives, according to new research from scientists at Weill Cornell Medicine. A study, published Feb. 5 in Cell, reveals a new twist in the complex relationship between humans and their associated microbes, and points the way toward novel therapies that could help combat a rising tide of drug-resistant pathogens.



