News
Lymphoma mutation yields super-competitive immune cells
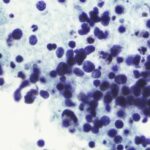 The key to understanding how the most aggressive lymphomas arise and resist current therapies may lie in mutations that disrupt a critical natural selection process among antibody-producing B cells, according to a multi-institutional preclinical study led by Weill Cornell Medicine investigators.
The key to understanding how the most aggressive lymphomas arise and resist current therapies may lie in mutations that disrupt a critical natural selection process among antibody-producing B cells, according to a multi-institutional preclinical study led by Weill Cornell Medicine investigators.
Scientists detail major mechanism lung cancers use to evade immune attack
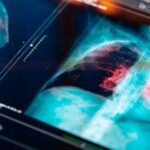 A protein commonly found at high levels in lung cancer cells controls a major immunosuppressive pathway that allows lung tumors to evade immune attack, according to a study led by researchers at Weill Cornell Medicine.
A protein commonly found at high levels in lung cancer cells controls a major immunosuppressive pathway that allows lung tumors to evade immune attack, according to a study led by researchers at Weill Cornell Medicine.
Common dietary fiber promotes allergy-like immune responses
New study finds that dietary inulin fiber alters the metabolism of certain gut bacteria, which in turn triggers what scientists call type 2 inflammation in the gut and lungs.
study finds that dietary inulin fiber alters the metabolism of certain gut bacteria, which in turn triggers what scientists call type 2 inflammation in the gut and lungs.
Study offers new insights into immune mechanisms of inflammatory disease
A new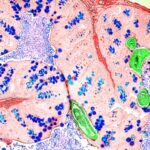 study suggests that a unique set of regulatory networks controlled by neurons in the gut may be viable targets for future drug therapies to combat chronic inflammatory diseases including asthma, allergy and inflammatory bowel disease.
study suggests that a unique set of regulatory networks controlled by neurons in the gut may be viable targets for future drug therapies to combat chronic inflammatory diseases including asthma, allergy and inflammatory bowel disease.
Pain-sensing gut neurons protect against inflammation
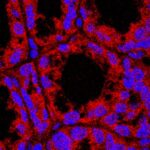 “A whole new approach”: Neurons that sense pain protects the gut from inflammation and associated tissue damage by regulating the microbial community living in the intestines, according to a study from Weill Cornell researchers.
“A whole new approach”: Neurons that sense pain protects the gut from inflammation and associated tissue damage by regulating the microbial community living in the intestines, according to a study from Weill Cornell researchers.
Undergrad publishes theory on immune dysfunction in space
 Rocky An ’23 proposes a theory that could solve the decades-old mystery of why astronauts’ immune systems become suppressed in space.
Rocky An ’23 proposes a theory that could solve the decades-old mystery of why astronauts’ immune systems become suppressed in space.
Cells help immune system tolerate friendly gut bacteria
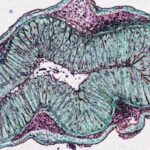 Immune cells called group 3 innate lymphoid cells play an essential role in establishing tolerance to symbiotic microbes that dwell in the human gastrointestinal tract, according to a study led by researchers at Weill Cornell Medicine.
Immune cells called group 3 innate lymphoid cells play an essential role in establishing tolerance to symbiotic microbes that dwell in the human gastrointestinal tract, according to a study led by researchers at Weill Cornell Medicine.
Study reveals new mode of triggering immune responses
Small proteins, called chemokines, that direct immune cells toward sites of infection can also form DNA-bound nanoparticles that can induce chronic, dysfunctional immune responses, according to a new study.
Center for Immunology connects Cornell strengths
 The center, with more than 120 faculty members, builds on the multidisciplinary nature of research into the immune system, with links between infection biology, vaccine development, genetics, genomics, malignancy and biomedical engineering.
The center, with more than 120 faculty members, builds on the multidisciplinary nature of research into the immune system, with links between infection biology, vaccine development, genetics, genomics, malignancy and biomedical engineering.


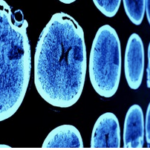 Li Gan studies how abnormal proteins and the body’s immune system drive Alzheimer’s disease.
Li Gan studies how abnormal proteins and the body’s immune system drive Alzheimer’s disease.
